What is ESMR Treatment?
ESMR was developed by Essen Institute of Cardiology, Essen University, Germany. The treatment is performed using a shock wave generator that is designed to address the unique.
Clinical-anatomical requirements of the chest cavity. A cardiac ultrasound imaging system is used to locate the treatment area and to map the exact position and extent of the ischemic zone. Shock waves are then delivered via the anatomical acoustic window to the treatment area under E.C.G. R-wave gating. Several treatment sessions are required to obtain optimal results.
The therapeutic effect of shock waves is attributed to the mechanical pressure and tension that the acoustic wave exerts on the cardiac tissue. Delivery of low intensity Shock waves causes sheer stress on the vessel wall. This stimulates angiogenesis related growth factors-( eNOS,VEGF and PCNA) initiating the angiogenesis process.
What are the Indications of ESMR?
Chronic chest pain (Refractory Angina) even after Bypass surgery or Angioplasty.
Having 70 % -100 % blockages in angiogram.
Re-blocks after Bypass surgery or Angioplasty.
Chronic Renal disease with heart arteries block.
High risk cases medically unfit for Bypass surgery/angioplasty.
Patients’ coronary anatomy not readily amenable for Bypass surgery.
Inability to perform angioplasty because the target lesion is inaccessible/ diffuse.
Patients not willing for surgical procedures/ angioplasty.
How is the treatment done?
During the treatment, the patient lies on a table. Echocardiography Imaging is performed to ensure precise applicator positioning. Focussed Low-Energy Shockwaves are then applied non invasively. This will create collaterals and improve blood flow to the targeted portions of the heart muscle.
The whole treatment duration is 20-30 minutes and the patient should undergo a series of nine treatment sessions during a nine week period.(3 treatments at first week of each month- 1st week , 5th week, 9th week). At each treatment session shockwaves are delivered under echocardiographic guidance.
Clinical benefits of ESMR
Patient would have no angina.
Episodes of angina would be less.
Patient need for anti anginal medications can be reduced/stopped.
Patient can return to work and can participate in their active life style once again.
Patient would be more energetic and confident and walk more distance without chest pain.
Currently in use in outpatient facilities around the world.
Artificial Heart
Dr Kamales Kumar Saha is one of the few surgeons in India who has undergone the Heartware 2 certified training in Berlin Heart Hospital in Germany. If you have been advised a heart transplant or a LVAD or artificial heart, contact Dr Kamales Kumar Saha for an opinion.
A ventricular assist device (VAD) is an electromechanical circulatory device that is used to partially or completely replace the function of a failing heart. The function of VADs differs from that of artificial cardiac pacemakers. Some VADs are intended for short term use, typically for patients recovering from heart attacks or heart surgery, while others are intended for long-term use (months to years and in some cases for life), typically for patients suffering from advanced congestive heart failure.
VADs are designed to assist either the right (RVAD) or left (LVAD) ventricle, or both at once (BiVAD). The type that is used depends primarily on the underlying heart disease and the pulmonary arterial resistance that determines the load on the right ventricle.
LVADs are most commonly used, but when pulmonary arterial resistance is high, RVAD may become necessary. Long term VADs are normally used to keep patients alive with a good quality of life while they wait for a heart transplantation (known as a "bridge to transplantation"). However, the newer LVADs are often used as destination therapy, meaning the patients live a good quality life with the LVAD and there is no need for them to undergo heart transplant.
In the last few years, VADs have improved significantly in terms of providing survival and quality of life among patients.
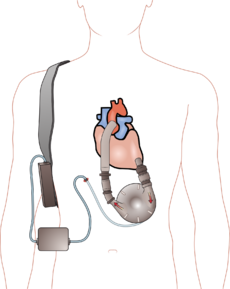
- A ventricular assist device has three parts:
- A pump the newer LVADS have very small powerful pumps which can be placed in the outer covering of your heart pericardium.
- An electronic controller. The controller is like a small computer that controls how the pump works.
- Batteries. The batteries are carried outside your body. They are connected to the pump with a cable that usually goes into your belly.
Valve Surgeries
The heart has four valves. Normally, these valves open to let blood flow through or out of your heart, and then shut to keep it from flowing backward. But sometimes they don't work properly.
- Blood can leak back through the valve in the wrong direction, which is called regurgitation
- When the valve doesn't open enough, which blocks blood flow, it is called stenosis.
- One of the valves, the mitral valve, sometimes has "floppy" flaps and doesn't close tightly. This is called mitral valve prolapse, and it's one of the most common heart valve conditions. Sometimes it causes regurgitation
Valve problems can be present at birth or caused by infections, heart attacks, or heart disease or damage. Some valve problems are minor and do not need treatment. Others might require medicine, medical procedures or surgery to repair or replace the valve.
Heart valve repair
Whenever possible a heart valve should be repaired rather than replaced. Heart valve repair leaves you with your normally functioning tissue, which resists infection more effectively, and you don't need to take blood-thinning medications after the surgery.
Depending on the condition using one of three surgeries are done.
-
Annuloplasty
In this procedure, your surgeon tightens the ring of tissue around the valve to help the valve flaps (leaflets) come together. Sometimes the surgeon implants a ring where the leaflets meet to make the valve opening smaller so the leaflets can come together.

-
Repair of structural support
In this procedure, the surgeon replaces or shortens the cords that support the valves (chordae tendineae and papillary muscles) to repair the structural support. When the cords and muscles are the right length, the valve leaflet edges meet and eliminate the leak.
-
Valve leaflet repair
In valve leaflet repair, the surgeon surgically separates cuts or pleats a valve flap (leaflet) to repair a floppy or prolapsed valve leaflet.

Part of a prolapsing mitral valve is removed to allow the valve to fully close and stop leaking.
Heart valve replacement
If the heart valve can't be repaired, the damaged valve will be replaced with an artificial (prosthetic) valve. Heart valve replacement are required if there is valve narrowing (stenosis) or severely damaged valves affected by calcium build up in the valves (calcification) or rheumatic disease. Mitral or aortic valves are the commonly replaced valves, but any heart valve can be replaced.
Heart valves can be replaced by:
-
Mechanical valves
Mechanical valves are made of synthetic materials, which are designed to last many years. However, blood tends to stick to mechanical valves and create blood clots. So blood-thinning medicines (anticoagulants) have to be taken for life.

-
Biological valves
Biological valves, or tissue valves, are made from animal tissue (xenograft) or taken from the human tissue of a donated heart (called an allograft or homograft). Sometimes, patients own tissue can be used for valve replacement (autograft/Ross procedure).
If you have a biological valve, usually there is no need to take blood-thinning medication. However, biological valves aren't as durable and are more costly as compared to mechanical valves, and eventually may need to be replaced. Surgeons mostly use biological valves in older patients or patients who cannot take blood thinning medications.
Maze Procedure
The Maze Procedure is surgery performed to treat atrial fibrillation. During the procedure, a number of incisions are made on the left and right atrium to form scar tissue, which does not conduct electricity and disrupts the path of abnormal electrical impulses. The scar tissue also prevents erratic electrical signals from recurring. After the incisions are made, the atrium is sewn together to allow it to hold blood and contract to push blood into the ventricle.
Although the atrium is sewn back together, the erratic electrical paths remain severed so that no unwanted electrical impulse can cross the incision. The result is what looks like a children's maze in which there is only one path that the electrical impulse can take from the SA node to the AV node. The atrium can no longer fibrillate, and sinus rhythm (the normal rhythm of the heart) is restored. Maze can be performed either through an open chest procedure or a minimally invasive procedure. Ninety percent of Maze surgeries are concomitant (done in conjunction) with other open chest surgery, such as coronary artery bypass grafting, mitral valve repair and/or valve replacement.
ECG Before MAZE
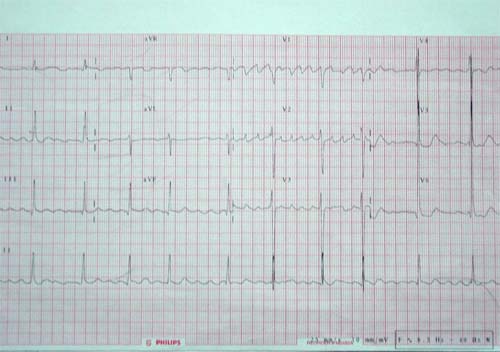
ECG After MAZE

Aortic Aneurysms Procedure
The aorta is the main artery carrying oxygen rich blood from the heart. In the chest, the aorta is called the thoracic aorta; in the abdomen, the abdominal aorta. Arteries are muscular tubes made up of 3 layers: the innermost layer (the intima), the middle layer (the media), and the outer layer (the adventitia). Abnormal enlargement or bulging of the aorta is not an unusual condition.
Bulges in an artery are classified as true aneurysm, false aneurysm, or dissection.
A true aneurysm involves all 3 layers of the blood vessel wall.
A false aneurysm of the artery is contained only by the 2 outer layers of the blood vessel wall and clot. This is a very fragile condition with high risk of bursting. False aneurysms are usually a result of an infection.
A related but different condition is aortic dissection. Dissection refers to a separation of the vessel wall, which allows blood to leak between the layers of the vessel. This further damages and weakens the vessel, placing it at much greater risk of bursting (rupture).
A ruptured abdominal aneurysm is a life-threatening event. Fortunately, not all aneurysms rupture right away. Many grow very slowly and cause no symptoms or problems for many years. When detected in time, most aneurysms can be electively repaired with an operation so they do not rupture. Most aneurysms occur in people aged 55 years or older and are 4 times more common in men than in women.
Medical Treatment
The treatment of this condition depends on your overall health, the size and location of the aneurysm, and whether you are having symptoms. Close monitoring of the aneurysm size is the proper treatment for most people.
Ultrasound/CT scan every 6-12 months is the usual monitoring strategy. This may vary by individual patient and health care provider.
Any abdominal aortic aneurysm larger than about 2 inches (5.5 cm) in size requires surgery to repair it. Aneurysms that are enlarging rapidly, causing symptoms, or showing signs of probably rupture (such as leaking) require immediate surgery. Delaying this surgery puts you at even greater risk of a rupture.
Thoracic aortic aneurysms may originate in either the ascending or descending aorta and, because of their closer proximity to the heart than abdominal aortic aneurysms, they have greater potential to harm the heart or create other problems related to the heart. Usually Any thoracic aortic aneurysm 6 cm or larger requires surgery, but in many cases smaller aneurysms are considered for surgery.
Open Surgery
Open surgery has long been the accepted treatment for aortic aneurysms of the chest or abdomen.
A large incision is made in the chest or the abdomen. Blood flow in the aorta is stopped by hooking the circulatory system to an outside pump (heart and lung machine). This machine circulates blood to the body and keeps the vital organs and other tissues oxygenated properly.
The damaged section of the aorta is removed and replaced by an artificial blood vessel (graft) made of cloth fabric. The graft is sewn in place. Usually a patient has to stay in the hospital for a week, and recovery time is at least 6 weeks.
Endovascular stenting
Treatment for these aneurysms has recently focused on procedures that are less invasive than conventional surgery.
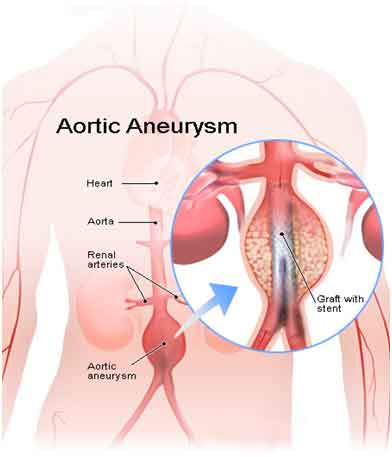
One widely used alternative is stenting of the aneurysm. A stent is a tiny metal and fabric device like a miniature scaffold. It is threaded through the blood vessel from a small incision, usually in the groin. A thin plastic tube called a catheter is used to thread the stent through the blood vessel. The stent is fastened to the inner vessel wall just above the level of weakened aortic wall to help support the blood vessel. This reduces strain on an existing aneurysm and can prevent a full-blown aneurysm from developing again. Stenting is not only much faster than conventional surgery, it offers much shorter recovery time and fewer complications.
The appropriate choice of procedure, open versus stenting, depends on many factors and is best determined by your surgeon.
Congenital corrections
With congenital heart defects, some part of the heart doesn’t form properly before birth. This changes the normal flow of blood through the heart.
There are many types of congenital heart defects. Some are simple, other heart defects are more complex.
Simple Congenital Heart Defects-Holes in the Heart (Septal Defects)
The septum is the wall that separates the chambers on left and right sides of the heart. The wall prevents blood from mixing between the two sides of the heart. Some babies are born with holes in the septum. These holes allow blood to mix between the two sides of the heart.
Atrial septal defect (ASD)
An ASD is a hole in the part of the septum that separates the atria—the upper chambers of the heart. The hole allows oxygen-rich blood from the left atrium to flow into the right atrium, instead of flowing into the left ventricle as it should. Many children who have ASDs have few, if any, symptoms.
ASDs can be small, medium, or large. Small ASDs allow only a little blood to leak from one atrium to the other. They don't affect how the heart works and don't need any special treatment. Many small ASDs close on their own as the heart grows during childhood.
Medium and large ASDs that need treatment can be repaired using a catheter procedure or open-heart surgery.
Ventricular septal defect (VSD)
A VSD is a hole in the part of the septum that separates the ventricles—the lower chambers of the heart. The hole allows oxygen-rich blood to flow from the left ventricle into the right ventricle, instead of flowing into the aorta and out to the body as it should.
VSDs can be small, medium, or large. Small VSDs don't cause problems and may close on their own. Medium VSDs are less likely to close on their own and may require treatment.
Large VSDs allow a lot of blood to flow from the left ventricle to the right ventricle. As a result, the left side of the heart must work harder than normal. Extra blood flow increases blood pressure in the right side of the heart and the lungs.
The heart’s extra workload can cause heart failure and poor growth. If the hole isn't closed, high blood pressure can scar the arteries in the lungs.
Doctors use open-heart surgery to repair VSDs.
Patent Ductus Arteriosus
Patent ductus arteriosus (PDA) is a fairly common heart defect that can occur soon after birth. In PDA, abnormal blood flow occurs between the aorta and the pulmonary artery.
Before birth, these arteries are connected by a blood vessel called the ductus arteriosus. This blood vessel is an essential part of fetal blood circulation. Within minutes or up to a few days after birth, the ductus arteriosus closes.
In some babies, however, the ductus arteriosus remains open (patent). The opening allows oxygen-rich blood from the aorta to mix with oxygen-poor blood from the pulmonary artery. This can strain the heart and increase blood pressure in the lung arteries.
A heart murmur might be the only sign of PDA. Other signs and symptoms can include shortness of breath, poor feeding and growth, tiring easily, and sweating with exertion.
PDA is treated with medicines, catheter-based procedures, and surgery. Small PDAs often close without treatment.
Narrowed Valves
Simple congenital heart defects also can involve the heart's valves. These valves control the flow of blood from the atria to the ventricles and from the ventricles into the two large arteries connected to the heart (the aorta and the pulmonary artery).
Valves can have the following types of defects:
- Stenosis (steh-NO-sis). This defect occurs if the flaps of a valve thicken, stiffen, or fuse together. As a result, the valve cannot fully open. Thus, the heart has to work harder to pump blood through the valve.
- Atresia (ah-TRE-ze-AH). This defect occurs if a valve doesn't form correctly and lacks a hole for blood to pass through. Atresia of a valve generally results in more complex congenital heart disease.
- Regurgitation (re-GUR-jih-TA-shun). This defect occurs if a valve doesn't close tightly. As a result, blood leaks back through the valve.
Severe pulmonary valve stenosis is treated with a catheter procedure.
Complex Congenital Heart Defect
Tetralogy of Fallot
It is a combination of four defects:
- Pulmonary valve stenosis.
- A large VSD.
- An overriding aorta. In this defect, the aorta is located between the left and right ventricles, directly over the VSD. As a result, oxygen-poor blood from the right ventricle can flow directly into the aorta instead of into the pulmonary artery.
- Right ventricular hypertrophy. In this defect, the muscle of the right ventricle is thicker than usual because it has to work harder than normal.
In tetralogy of Fallot, not enough blood is able to reach the lungs to get oxygen, and oxygen-poor blood flows to the body.
Babies and children who have tetralogy of Fallot have episodes of cyanosis, which can be severe. In the past, when this condition wasn't treated in infancy, older children would get very tired during exercise and might faint. Tetralogy of Fallot is repaired in infancy now to prevent these problems.
Tetralogy of Fallot must be repaired with open-heart surgery, either soon after birth or later in infancy. The timing of the surgery will depend on how narrow the pulmonary artery is.
For more information visit http://www.ncbi.nlm.nih.gov/pubmedhealth/PMH0062980/
 Check How Healthy Your Heart Is?
Check How Healthy Your Heart Is?